About Us
InAce Pharma Consulting LLP
At InACE,
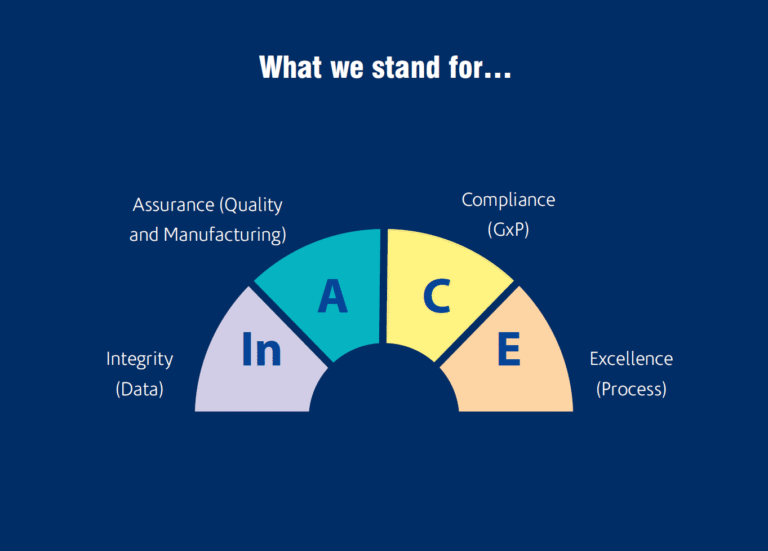
We are a team of experts specialized in Quality Control, Quality Assurance, Manufacturing, Engineering, Project Management and Regulatory.
We are committed to being your trusted partner in achieving and sustaining GMP compliance through rigorous audits, impactful training, and a strong quality culture.
We support our clients in implementing sustainable solutions, that build long-term compliance and resilience to meet global standards like US-FDA, EU-GMP, MHRA, and other global agencies.
We empower your teams to perform with confidence and clarity — making your operations audit-ready at all times.
Beyond compliance, we at InACE bring proven expertise in process excellence and operational improvement, helping clients enhance manufacturing efficiency, optimize costs, and enhance organizational capability.
We believe in a collaborative, hands-on approach tailored to your specific needs — strengthening systems, building people, and making processes more robust and competitive.
We work either on retainership basis or on project basis, depending upon the assignment.
We look forward to partnering with you to build safer products, stronger operations, and a more resilient business.
Meet Our Team!

Balaji Krishnan
Director
QS, Micro & Sterility Assurance

Padmini Dalal
Director
Quality Systems & Compliance

Rajan Ramalingam
Director
International Affairs

Vijaya Sheoraman
Director
Operations & Finance

Shital Sadalge
Senior Advisor
Manufacturing

Dinesh Nambiar
Senior Advisor
Pharma & Consumer Products

Dilip Degwekar
Advisor
Inspection Readiness

Aarti Godbole
Consultant
Microbiology

Arra Srikar
Consultant
Quality Systems

Ramesh Vaidyanathan
Consultant
Analytical & Validations